⚡ The Power Problem in Implantable Medicine
For decades, implantable medical devices have been constrained by a fundamental limitation: batteries. Pacemakers, neurostimulators, cochlear implants, and drug pumps all require surgical replacement when batteries deplete—typically every 5-10 years. Each replacement surgery carries risks: infection, bleeding, device damage, and patient discomfort. For deep-brain stimulators or spinal cord implants, replacement surgery is particularly invasive.
Wireless power transfer promises to eliminate this limitation. By continuously powering implants through external transmitters, devices could operate indefinitely without battery replacement. The vision is transformative: lifetime neurostimulators, permanent glucose monitors, and implantable sensors that never require surgical intervention.
But wireless power transfer through human tissue introduces critical safety challenges. The same electromagnetic fields that deliver energy can also heat tissue, stimulate nerves, and potentially cause long-term biological effects. Two primary technologies compete for implant powering: inductive coupling (magnetic fields) and radio frequency (RF) energy transfer (electromagnetic waves). Each has distinct power transfer characteristics, efficiency profiles, and safety limits.
This article examines the safety boundaries of wireless-powered implants — the physical limits, regulatory standards, and design trade-offs that determine whether a device can be powered safely for a lifetime.
🧬 The Biophysics of Energy Transfer
Understanding safety limits requires understanding how electromagnetic energy interacts with biological tissue.
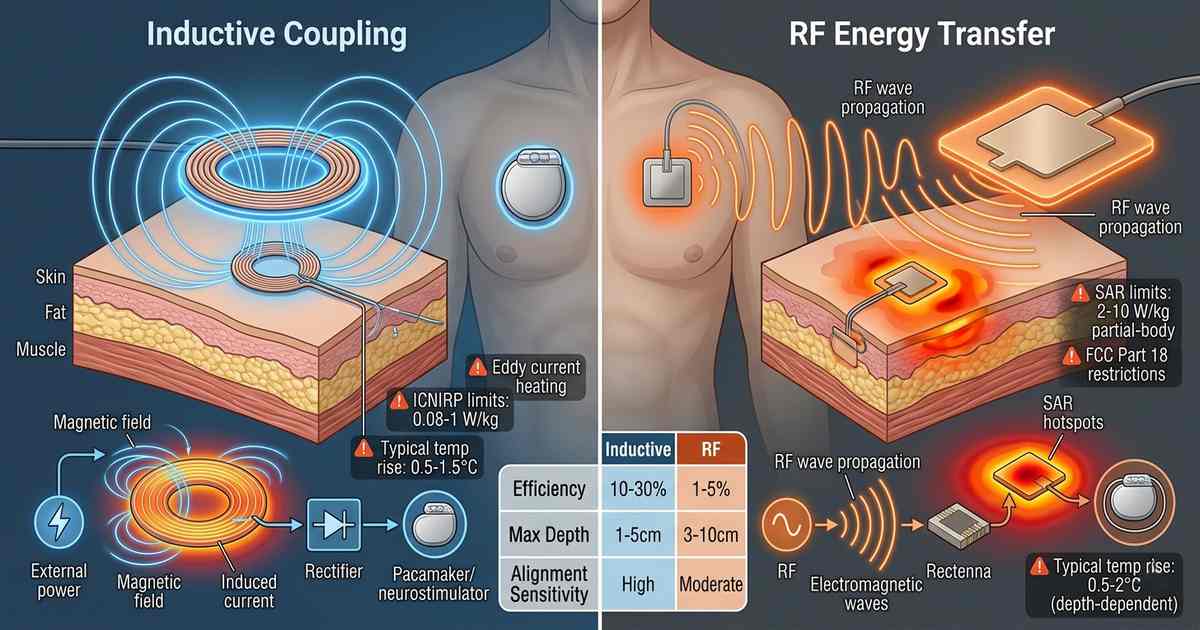
🔄 Inductive Coupling
Inductive power transfer uses magnetic fields generated by an external coil to induce current in an implantable coil. The operating frequencies typically range from 100 kHz to 10 MHz. At these frequencies, the human body is largely transparent to magnetic fields — tissue does not absorb significant energy from magnetic fields alone.
The safety concern with inductive coupling is primarily eddy current heating. The alternating magnetic field induces circulating currents in conductive tissue, generating heat through resistive losses. The heating effect is proportional to frequency squared and tissue conductivity. At frequencies below 1 MHz, eddy current heating is minimal; at frequencies approaching 10 MHz, it becomes significant.
📡 RF Energy Transfer
RF energy transfer uses electromagnetic waves — typically in the ISM bands at 915 MHz, 2.45 GHz, or higher — to transmit power to an implantable antenna or rectenna (rectifying antenna). RF waves are absorbed by tissue, with absorption depending on frequency, tissue type, and polarization.
The safety concern for RF transfer is specific absorption rate (SAR) — the rate at which tissue absorbs electromagnetic energy, measured in watts per kilogram (W/kg). RF energy heats tissue directly through dielectric absorption. Unlike inductive coupling, which concentrates fields near the coils, RF fields can penetrate deeply, potentially heating large tissue volumes.
🔌 Inductive Power Transfer: Safety Limits
📜 Regulatory Standards
Inductive implant systems fall under ICNIRP (International Commission on Non-Ionizing Radiation Protection) guidelines and IEEE C95.1 standards for time-varying magnetic fields. For frequencies below 100 kHz, the primary limit is based on induced electric fields in tissue — typically 0.02-0.1 V/m in the heart and central nervous system.
For frequencies from 100 kHz to 10 MHz (the range most inductive implants use), limits become more restrictive:
- Occupational exposure: 0.5-5 W/kg SAR (frequency-dependent)
- General public exposure: 0.08-1 W/kg SAR (frequency-dependent)
These limits are derived from thermal thresholds: exposure should not raise tissue temperature more than 1-2°C above baseline.
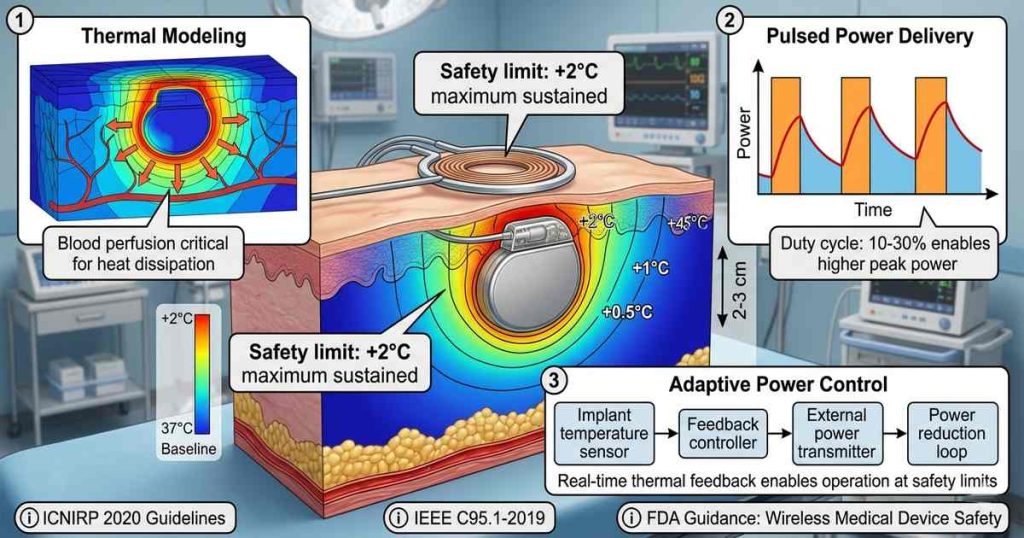
🌡️ Thermal Safety: The 2°C Threshold
The fundamental safety limit for all wireless implant powering is tissue heating. Neural tissue is particularly sensitive; temperature increases above 1°C can affect neuronal function. Sustained temperature increases above 2°C can cause tissue damage.
For inductive systems, thermal modeling must account for:
- Coil proximity: Implant coils are typically within 1-5 cm of the external coil
- Tissue conductivity: Muscle and blood have higher conductivity than fat or bone
- Blood perfusion: Well-perfused tissues (heart, liver) dissipate heat more effectively
A typical inductive system delivering 100 mW to an implant generates approximately 500 mW of power dissipation in the external coil and 100-200 mW of eddy current heating in tissue. With proper coil design and positioning, temperature rise can be maintained below 1°C.
🏥 Implant Heating Constraints
The implant itself generates heat. Rectification, regulation, and device operation convert some received power to heat. For a 50 mW implant (typical for neurostimulators), thermal dissipation of 10-20 mW concentrated in a small volume can raise local temperature by 2-3°C if not properly managed.
Design solutions include:
- Distributed heating: Spreading power dissipation across larger surface area
- Thermal conduction: Using device casing as heat spreader
- Pulsed operation: Delivering power in short bursts rather than continuously
📡 RF Energy Transfer: Safety Limits
📊 SAR Limits and Tissue Penetration
RF energy transfer operates under stricter SAR limits. IEEE C95.1-2019 sets:
- Whole-body SAR: 0.4 W/kg (occupational), 0.08 W/kg (general public)
- Partial-body SAR: 10 W/kg (averaged over 10 g of tissue)
- Spatial peak SAR: 10 W/kg for extremities, 2 W/kg for head and torso
For implant powering, the relevant limit is partial-body SAR — the energy absorbed in the volume around the implant. A 100 mW RF power transfer system operating at 2.45 GHz might produce local SAR of 5-20 W/kg within 1 cm of the implant antenna, exceeding limits unless power levels are reduced.
📈 Frequency-Dependent Absorption
Tissue absorption varies dramatically with frequency:
| Frequency | Penetration Depth (muscle) | Primary Absorption | 100 kHz >1 m Minimal 1 MHz 50-100 cm Eddy currents 100 MHz 5-10 cm Dielectric 915 MHz 3-5 cm Dielectric (surface) 2.45 GHz 1-2 cm Dielectric (surface)
|---|
Higher frequencies deliver power more efficiently to small implants but concentrate absorption near the surface. For deep implants (e.g., pacemakers, deep-brain stimulators), RF frequencies above 1 GHz require high power levels to penetrate tissue, increasing surface SAR.
🎯 Antenna Design and Hot Spots
RF implant antennas create localized hot spots — regions of high SAR near the antenna structure. A small implant antenna (1-2 cm) can produce SAR values 10-100× higher than surrounding tissue. Even with low total power, these hot spots can exceed safety limits.
Mitigation strategies include:
- Broadband antennas: Spreading energy across frequency range
- Directional patterns: Shaping radiation away from sensitive tissues
- Implant encapsulation: Using biocompatible materials with thermal conductivity
⚖️ Comparative Analysis: Inductive vs RF
🔋 Power Transfer Efficiency
| Metric | Inductive (1-10 MHz) | RF (915 MHz-2.45 GHz) | Efficiency (1-5 cm depth) 10-30% 1-5% Maximum deliverable power 500 mW+ 50-100 mW Distance sensitivity High (d⁻³ to d⁻⁶) Moderate (d⁻²) Alignment tolerance Critical Moderate
|---|
Inductive systems achieve higher efficiency at shallow depths but drop off rapidly with distance and misalignment. RF systems maintain more consistent power with distance but at lower efficiency. For deep implants (>5 cm), RF may be the only feasible option despite lower efficiency.
🌡️ Thermal Performance
| Metric | Inductive | RF | Primary heating mechanism Eddy currents (volume) Dielectric absorption (local) Thermal management Coil placement, duty cycle SAR distribution, hot spot control Typical temperature rise 0.5-1.5°C 0.5-2°C (challenging at depth)
|---|
Inductive heating is more distributed, making thermal management simpler. RF heating is highly localized, requiring careful antenna design and power budgeting.
⚖️ Regulatory Compliance
Inductive systems operate in bands with less restrictive SAR limits. The FCC permits higher power levels for inductive chargers (e.g., Qi wireless charging at 100-300 kHz) than for RF power transfer.
RF systems face stricter limits:
- FCC Part 18 (ISM): Limited to 10-50 W EIRP for industrial, scientific, medical use
- Medical device exemptions: Some allowances for implant powering, but case-by-case
- Europe (ETSI EN 303 417): Similar restrictions
🔮 Emerging Safety Considerations
⏱️ Long-Term Exposure
Most safety limits are derived from short-term thermal effects. The long-term biological effects of continuous electromagnetic exposure remain less understood. For implants designed to operate for decades, the cumulative exposure is unprecedented.
Key research areas include:
- Cellular effects: Potential for non-thermal biological effects at chronic exposure
- Tissue adaptation: Whether tissue responds to chronic heating with adaptive mechanisms
- Device-tissue interface: Long-term effects on encapsulation and biocompatibility
🔁 Multiple Implants
As patients receive multiple implants — pacemaker, neurostimulator, glucose monitor — the cumulative electromagnetic exposure increases. Safety standards currently consider single-device exposure; multi-device scenarios are not well addressed.
🎛️ Dynamic Power Control
Modern wireless implant systems incorporate feedback loops that adjust power based on implant needs and tissue temperature. Real-time thermal monitoring enables operation at safety limits without conservative static power caps. Implantable temperature sensors can detect heating and trigger power reduction before tissue exceeds safety thresholds.
🏥 Clinical Implementations
🔄 Inductive Systems
Medtronic’s implantable neurostimulators use inductive charging at 100-500 kHz. External chargers deliver 100-200 mW to implants at 1-2 cm depth with temperature rise <1°C. Patients charge for 1-2 hours weekly — intermittent exposure well within safety limits.
Cochlear implants use inductive transmission for both power and data. Transcutaneous coils at 5-10 MHz deliver 50-100 mW across skin with heating managed through pulsed operation and careful coil alignment.
📡 RF Systems
Wireless pacemakers (e.g., EBR Systems’ WiSE CRT system) use RF energy at 915 MHz to power left ventricular electrodes. The system delivers 10-30 mW to deep cardiac tissue with SAR managed through antenna design and duty cycling. Clinical trials demonstrate acceptable safety profiles with temperature rise <0.5°C.
Wireless retinal implants (e.g., Pixium Vision) use RF at 2.45 GHz to power epiretinal electrodes. The system operates at very low power (<10 mW) with pulsed delivery, maintaining SAR within safe limits despite the implant’s proximity to brain tissue.
⚖️ The Efficiency-Safety Trade-Off
The fundamental engineering challenge is balancing power delivery with safety. Higher power enables more capable implants — more stimulation channels, higher sampling rates, wireless data transmission — but increases thermal risk.
The design space is constrained by:
- Implant depth: Shallow implants favor inductive; deep implants require RF
- Power requirement: High-power devices (>100 mW) favor inductive
- Duty cycle: Continuous operation demands tighter safety margins
- Anatomical location: Proximity to sensitive tissues (brain, heart, eyes) imposes stricter limits
🚀 Future Directions
🔄 Mid-Frequency Hybrid Systems
Systems operating at 10-100 MHz — between traditional inductive and RF bands — offer potential compromises. At these frequencies, both inductive and dielectric coupling contribute, enabling efficiency gains without the safety challenges of higher frequencies.
🤖 Adaptive SAR Management
Real-time SAR monitoring and power adjustment can push systems closer to safety limits without exceeding them. Machine learning models that predict temperature rise based on tissue properties and implant position enable safe operation at higher average power.
💎 Implant Materials
New materials with higher thermal conductivity (diamond, silicon carbide) can spread heat more effectively, increasing safe power limits. Thermoelectric materials that convert waste heat to electrical power could recapture some dissipation energy.
📜 Regulatory Evolution
As wireless implants become more common, regulatory frameworks are evolving. The FDA’s guidance on wireless medical devices increasingly addresses chronic exposure, and industry standards (AAMI/ISO 14117) are incorporating wireless power safety requirements.
📌 Conclusion
Wireless-powered medical implants promise to eliminate the battery replacement surgeries that have long constrained implantable medicine. But realizing this promise requires navigating complex safety limits that balance power delivery against tissue heating.
Inductive coupling offers higher efficiency and simpler thermal management for shallow implants, operating safely within established regulatory limits. RF energy transfer enables deeper implants at the cost of lower efficiency and more challenging SAR management. Both approaches have achieved clinical success, with temperature rises maintained below 1°C — well within the 2°C safety threshold.
The future lies not in choosing one technology over the other but in optimizing each for specific applications and increasingly integrating both. Hybrid systems, adaptive power control, and improved thermal management will push the boundaries of what’s possible while maintaining the safety margins that patients and regulators demand.
For patients, the implications are profound: neurostimulators that never need replacement, glucose monitors that last a lifetime, and implantable sensors that provide continuous health data without surgical intervention. The power is wireless. The safety is proven. The era of permanent implantable devices has begun.