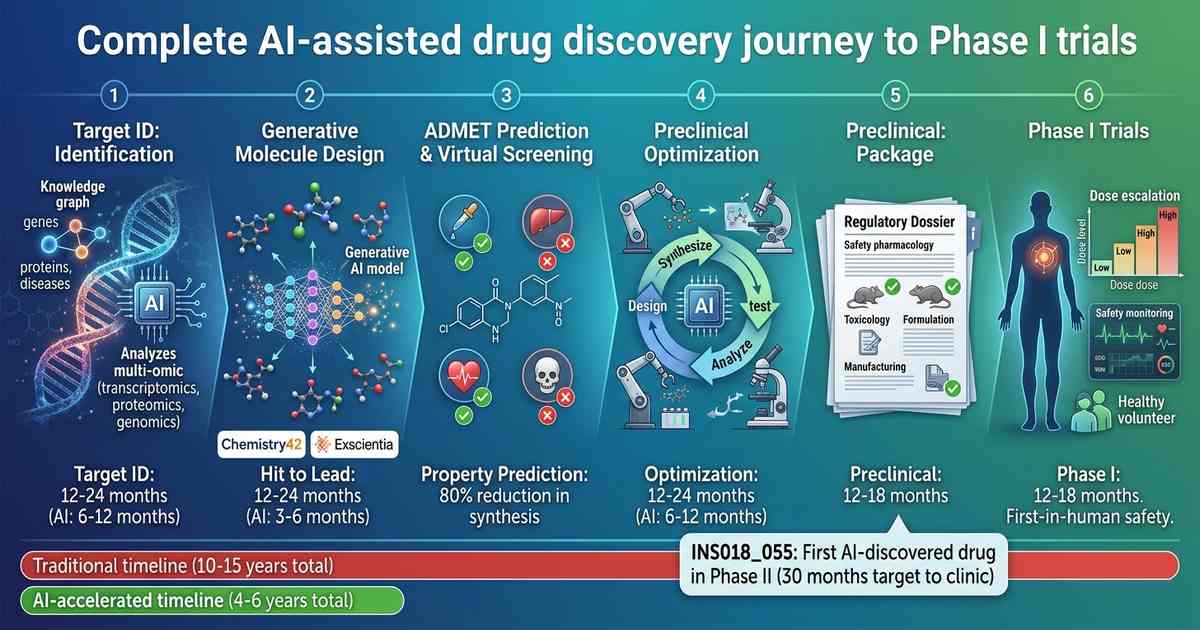
The Crisis in Pharmaceutical R&D
Developing a new drug is one of the most expensive, time-consuming, and failure-prone endeavors in human history. The numbers are staggering: an average of $2.6 billion in research and development costs, 10 to 15 years from initial discovery to market approval, and a 90% failure rate in clinical trials. For every drug that reaches patients, nine others fail somewhere along the pipeline—often after hundreds of millions of dollars have already been invested.
This crisis in pharmaceutical productivity has persisted for decades, defying technological advances that have transformed virtually every other industry. The cost per approved drug has doubled approximately every nine years since 1950—a phenomenon known as Eroom’s Law (Moore’s Law spelled backward). Despite exponential increases in R&D spending, the number of new drugs approved per billion dollars invested has fallen by 80% since 1960.
Artificial intelligence is now challenging this trajectory. AI-assisted drug discovery promises to compress timelines, reduce costs, increase success rates, and ultimately deliver better medicines to patients faster. The technology has moved from academic curiosity to industry standard in just a few years, with multiple AI-discovered molecules now in human clinical trials. This article traces the AI drug discovery pipeline from the earliest stages of target identification through the critical milestone of Phase I trials.
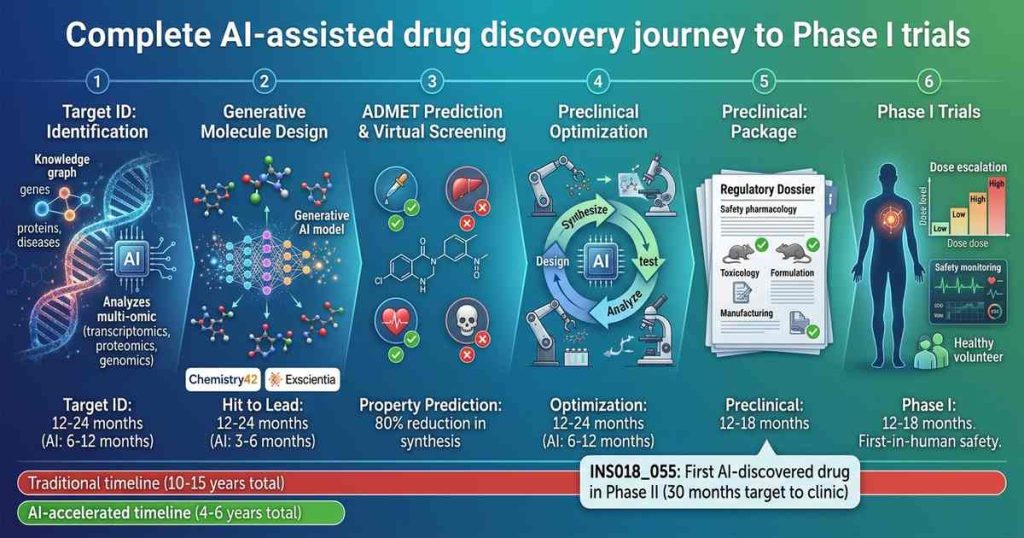
The Traditional Drug Discovery Pipeline: A History of Inefficiency
Understanding AI’s impact requires understanding what it replaces. Traditional drug discovery follows a linear, high-friction path:
Target Identification and Validation (2-4 years): Researchers identify a biological target—typically a protein implicated in disease—and validate that modulating it produces therapeutic benefit. This phase relies on academic literature, genetic studies, and laborious experimental validation.
Hit Discovery (1-2 years): Scientists screen libraries of compounds (often hundreds of thousands to millions) against the target to identify “hits”—molecules that show initial activity. High-throughput screening (HTS) can test millions of compounds but requires massive infrastructure and yields mostly false positives.
Lead Optimization (2-4 years): Promising hits are iteratively modified to improve potency, selectivity, pharmacokinetics, and safety. Medicinal chemists synthesize and test thousands of analogs in a slow, expensive cycle averaging $10,000 to $50,000 per compound tested.
Preclinical Development (1-2 years): Optimized leads undergo animal studies to assess safety, efficacy, and dosing. Most compounds fail here due to toxicity or poor pharmacokinetics.
Phase I Trials (1-2 years): First-in-human studies assess safety, tolerability, and pharmacokinetics in 20-100 healthy volunteers. Only about 10% of drugs entering Phase I eventually receive approval.
The cumulative inefficiency is staggering. For every drug that reaches patients, thousands of molecules are synthesized and tested, hundreds of animals are used, and billions of dollars are spent on failures.
AI in Target Identification: Mining the Biomedical Literature
The earliest stages of drug discovery are being transformed by AI’s ability to synthesize vast quantities of biomedical knowledge.
Literature Mining and Knowledge Graphs
The scientific literature grows exponentially—over 1 million new biomedical papers are published annually. No human researcher can keep pace. AI systems like BenevolentAI’s knowledge graph and Insilico Medicine’s PandaOmics ingest this corpus, extracting relationships between genes, proteins, diseases, pathways, and existing drugs.
These systems build comprehensive maps of biological knowledge, identifying novel connections that human researchers might miss. In 2020, BenevolentAI used its platform to identify baricitinib as a potential treatment for COVID-19 by recognizing that the drug’s mechanism of action (JAK inhibition) might modulate the inflammatory response central to severe COVID-19. The drug received emergency use authorization within months—a discovery that would have taken years through traditional literature review.
Multi-Omics Integration
AI platforms integrate data across biological scales: genomics (DNA), transcriptomics (RNA expression), proteomics (protein abundance), metabolomics (metabolites), and clinical data. By identifying patterns across these layers, AI can distinguish disease-relevant targets from bystanders and identify patient subgroups most likely to respond.
Insilico Medicine’s PandaOmics analyzes multi-omic datasets to identify novel targets for diseases with few existing treatments. In 2023, the platform identified a novel target for idiopathic pulmonary fibrosis (IPF) that had not been previously implicated in the disease. The target is now being pursued in preclinical development.
Causal Inference and Genetic Validation
Traditional target identification often identifies correlations rather than causal drivers. AI systems incorporating genetic data—particularly human genetics from genome-wide association studies (GWAS)—can prioritize targets with human genetic validation. A target supported by human genetics is significantly more likely to succeed in clinical trials than one identified through cell-based screens alone.
Recursion Pharmaceuticals uses its AI platform to integrate CRISPR screens (gene knockouts) with phenotypic imaging data, systematically identifying genes whose perturbation produces disease-relevant cellular changes. This approach generates causal hypotheses rather than correlative associations.
Generative AI for Molecule Design
Perhaps the most transformative AI application in drug discovery is generative chemistry—using AI to design novel molecules with desired properties.

From Screening to Generation
Traditional drug discovery relies on screening: testing existing compounds or libraries to find hits. This approach is inherently limited by the compounds available for testing. The chemical space of potential drug-like molecules is estimated at 10⁶⁰—vastly larger than all molecules ever synthesized. Screening explores only a microscopic fraction.
Generative AI flips the paradigm. Instead of screening what exists, AI models generate molecules optimized for specific properties. These models—often based on variational autoencoders (VAEs), generative adversarial networks (GANs), or transformers—learn the grammar of chemistry and can propose novel structures that satisfy multiple constraints simultaneously.
Insilico Medicine’s Chemistry42
The most mature generative chemistry platform is Insilico Medicine’s Chemistry42, an end-to-end AI system that designs novel molecules optimized for potency, selectivity, safety, and pharmacokinetics. The platform integrates multiple generative algorithms, allowing medicinal chemists to specify design objectives and receive novel molecule proposals within days rather than years.
In 2023, Insilico announced that a molecule discovered entirely by AI—ISM001-055 for idiopathic pulmonary fibrosis—had entered Phase II clinical trials. The journey from target identification to Phase I completion took just 30 months and cost a fraction of traditional development. The molecule was designed, synthesized, and tested in animals entirely under AI guidance. This milestone demonstrated that AI could not just accelerate individual steps but compress the entire discovery pipeline.
Exscientia and the First AI-Designed Drug in Human Trials
Exscientia achieved the first AI-designed drug to enter human trials: DSP-1181 for obsessive-compulsive disorder, developed in collaboration with Sumitomo Dainippon Pharma. The molecule was designed using Exscientia’s AI platform, which generated novel compounds optimized for serotonin 5-HT1A receptor agonism. From project initiation to candidate selection took approximately 12 months—roughly one-fifth the traditional timeline.
While DSP-1181 ultimately did not progress past Phase I (highlighting that AI does not guarantee success), it demonstrated that AI-designed molecules could meet the stringent safety and manufacturing standards required for human trials.
Structure-Based Design with AlphaFold and Beyond
The 2020 breakthrough of AlphaFold—DeepMind’s AI system predicting protein structures from amino acid sequences—has transformed structure-based drug design. For the first time, researchers can generate accurate protein structures for targets without experimental structural data. AlphaFold has predicted structures for nearly all known proteins, creating a comprehensive atlas that structure-based design tools can exploit.
AI systems now integrate AlphaFold predictions with generative chemistry, designing molecules optimized to fit predicted binding pockets. Generate:Biomedicines and Genesis Therapeutics are among companies building platforms that combine protein structure prediction with generative molecule design, creating a unified pipeline from target sequence to novel molecule.
ADMET Prediction: Reducing Late-Stage Failures
The majority of drug candidates fail due to poor ADMET properties—Absorption, Distribution, Metabolism, Excretion, and Toxicity. Traditionally, these properties are evaluated through animal studies late in discovery, after significant investment in synthesis and optimization.
AI-based ADMET prediction models can evaluate thousands of virtual compounds for drug-like properties before any synthesis occurs. Models trained on large datasets of historical compounds (like the PubChem and ChEMBL databases) predict properties including:
- Solubility: Critical for oral absorption
- Permeability: Ability to cross cell membranes
- Metabolic stability: Resistance to liver enzymes
- CYP inhibition: Risk of drug-drug interactions
- hERG liability: Risk of cardiac arrhythmia
- Toxicity signals: Predictions of organ toxicity
By eliminating compounds with poor predicted properties early, AI reduces synthesis costs, accelerates timelines, and improves the probability that candidates entering animal studies will succeed.
Schrödinger (not to be confused with the physicist) has developed industry-standard physics-based and AI-based platforms for property prediction. Their software is used by virtually all major pharmaceutical companies to prioritize compounds for synthesis.
De Novo Synthesis Planning: From Design to Lab
Designing a molecule is meaningless if it cannot be synthesized. AI systems now integrate synthesis planning, proposing synthetic routes that are feasible with available chemistry.
Chematica (acquired by Merck) and Synthia (developed by MiliporeSigma) use AI to predict retrosynthetic pathways—breaking target molecules into available building blocks. These systems have been trained on millions of published chemical reactions and can propose synthetic routes that human chemists might miss. Integration of synthesis planning with generative design ensures that AI-proposed molecules are not just theoretically optimal but practically synthesizable.
Preclinical Optimization: The AI-Accelerated Loop
The traditional medicinal chemistry cycle—design, synthesize, test, analyze, redesign—is the central bottleneck in drug discovery. Each cycle takes weeks to months. AI shortens this cycle in multiple ways:
Virtual Screening: Instead of synthesizing all candidate molecules, AI models predict which are most promising. Synthesis focuses only on high-probability candidates.
Active Learning: AI models learn from each experiment, updating predictions based on new data. The model improves with each cycle, requiring fewer experiments to reach optimization.
Multi-Parameter Optimization: Traditional optimization often optimizes one property at a time (potency first, then selectivity, then pharmacokinetics). AI models optimize all properties simultaneously, identifying molecules that balance multiple objectives.
Automated Synthesis: Emerging platforms like Emerald Cloud Lab and Arctoris integrate AI design with robotic synthesis, automating the design-synthesize-test loop. While still in early stages, these platforms hint at a future where the discovery cycle runs days rather than months.
The AI-Enabled Preclinical Package
For a candidate to enter Phase I trials, regulators require a comprehensive preclinical package demonstrating safety, efficacy, and manufacturing quality. AI contributes to each component:
Safety Pharmacology: AI models predict cardiovascular, respiratory, and central nervous system safety risks before animal studies.
Toxicology: Machine learning models trained on historical toxicology data identify potential organ toxicity signals, enabling early elimination of problematic compounds.
Formulation Development: AI predicts optimal formulations for bioavailability and stability, reducing the trial-and-error traditionally required.
Manufacturing Process: AI optimizes synthetic routes for scalability, cost-effectiveness, and environmental impact.
Phase I: AI-Optimized First-in-Human Trials
When an AI-discovered molecule enters Phase I trials, AI continues to contribute to trial design and analysis.
Patient Selection
While Phase I traditionally uses healthy volunteers, AI can identify patient subgroups most likely to show early efficacy signals. For oncology and rare diseases, AI-powered analysis of patient records can identify volunteers with the target biology, enabling earlier efficacy assessment.
Dosing Optimization
AI models incorporating preclinical pharmacokinetic data predict optimal human starting doses, reducing the risk of subtherapeutic dosing or unexpected toxicity. During the trial, Bayesian adaptive designs allow real-time dose adjustment based on emerging safety and pharmacokinetic data.
Biomarker Discovery
AI analysis of preclinical and early clinical data identifies biomarkers of target engagement and efficacy. These biomarkers become critical for patient selection in later-phase trials and for regulatory approval.
Real-World Evidence Integration
AI platforms integrate electronic health records, genomic databases, and real-world evidence to contextualize Phase I results. A molecule that shows early safety signals can be compared against outcomes in patients with similar characteristics who received standard care, providing preliminary evidence of potential benefit.
Case Studies: AI-Discovered Molecules in the Clinic
INS018_055 (Insilico Medicine)
Target: Idiopathic pulmonary fibrosis (IPF)
Discovery timeline: 18 months to preclinical candidate
Phase I completion: 2023
Status: Phase II ongoing
INS018_055 is the first entirely AI-discovered and AI-designed drug to enter human trials. Insilico’s platform identified a novel target (undisclosed) using PandaOmics, designed molecules using Chemistry42, and predicted properties using proprietary ADMET models. The molecule entered Phase I in 2022 and successfully completed safety assessment in 2023. Phase II efficacy trials are currently underway—a potential landmark for AI-driven drug development.
REC-994 (Recursion Pharmaceuticals)
Target: Cerebral cavernous malformation (CCM)
Discovery approach: AI-enabled phenotypic screening
Phase I completion: 2023
Status: Phase II ongoing
Recursion’s platform uses automated microscopy and AI image analysis to screen compounds for phenotypic effects in disease-relevant cell models. REC-994 was identified through this platform and has now entered Phase II for CCM—a rare disease with no approved treatments.
EXS-21546 (Exscientia)
Target: A2A receptor antagonist (immuno-oncology)
Discovery timeline: 8 months to candidate
Phase I initiation: 2022
Status: Phase I/II ongoing (with Evotec)
EXS-21546 was designed by Exscientia’s AI platform to be a highly selective A2A receptor antagonist for cancer immunotherapy. The molecule was advanced through Phase I and into a Phase I/II combination study with pembrolizumab.
Challenges and Limitations
Despite remarkable progress, AI-assisted drug discovery faces significant challenges:
Data Scarcity and Quality
AI models require large, high-quality datasets. Drug discovery data is often proprietary, fragmented, and inconsistent across sources. Public datasets like ChEMBL and PubChem, while valuable, represent only a fraction of all drug discovery data. Models trained on biased or incomplete data may generate biased predictions.
Interpretability
Regulators and clinicians require understanding of why a drug works and what risks it carries. Many AI models—particularly deep learning systems—operate as “black boxes,” making it difficult to explain predictions. Emerging techniques in interpretable AI aim to address this, but significant work remains.
Biological Complexity
Even the most sophisticated AI models cannot fully capture the complexity of human biology. A molecule that looks perfect in silico may fail in animals or humans due to unmodeled interactions. AI reduces but does not eliminate the need for empirical testing.
Regulatory Uncertainty
Regulatory frameworks evolved for traditional drug discovery. How agencies like the FDA and EMA will evaluate AI-discovered drugs remains evolving. While multiple AI-discovered molecules have entered trials, questions remain about how much AI-generated data can substitute for traditional experiments.
Integration with Existing Workflows
Pharmaceutical companies have deeply entrenched workflows, infrastructure, and culture. Integrating AI tools requires not just technology but organizational change. Many companies are building internal AI capabilities, but integration remains a work in progress.
The Future: Toward Fully AI-Driven Discovery
The trajectory of AI-assisted drug discovery points toward increasing autonomy:
Generative AI for Biologics: While most AI drug discovery has focused on small molecules, generative AI is increasingly applied to biologics—antibodies, peptides, and gene therapies. Platforms like Absci and BigHat Biosciences use AI to design antibodies optimized for binding, developability, and manufacturability.
Automated Laboratories: The integration of AI design with robotic synthesis and testing creates closed-loop discovery platforms. Companies like Zymergen (acquired by Ginkgo Bioworks) and Culture Biosciences are building automated biology platforms that can execute the design-build-test-learn cycle without human intervention.
Digital Twins for Clinical Trials: AI-created “digital twins”—virtual representations of individual patients—may enable virtual trials that predict real-world outcomes. These models, trained on historical trial data and real-world evidence, could optimize trial design, identify patient subgroups, and predict safety signals before trials begin.
Generative Biology: Beyond drug design, AI is learning to generate entirely novel biological entities—new proteins, pathways, and even organisms designed for therapeutic purposes. Profluent and Generate:Biomedicines are pioneering generative biology approaches that design proteins not found in nature.
Conclusion: A New Paradigm Emerges
The transformation of drug discovery by AI is not a future promise—it is happening now. From target identification to Phase I trials, AI tools are compressing timelines, reducing costs, and enabling approaches that were impossible a decade ago.
The first generation of AI-discovered molecules in clinical trials represents proof of concept. The next generation—currently in discovery pipelines—will determine whether AI can not just accelerate but fundamentally improve the probability of success. If AI can move the needle on the 90% clinical failure rate, the impact on patients, healthcare systems, and pharmaceutical economics will be profound.
The crisis in pharmaceutical productivity that defined the last half-century is being challenged by a new paradigm. AI-assisted drug discovery does not eliminate the need for rigorous science, careful experimentation, and regulatory oversight. But it promises to make those activities dramatically more efficient, more rational, and ultimately more successful.
For patients waiting for treatments, for researchers seeking new targets, and for an industry seeking to restore its productivity, AI-assisted drug discovery represents the most promising path forward. The molecules being designed today will become the medicines of tomorrow—faster, cheaper, and with higher probability of reaching the patients who need them.