Remote cardiac monitoring wearables have transformed cardiovascular care. From smartwatches detecting atrial fibrillation to implantable loop recorders tracking arrhythmias over years, these devices offer unprecedented visibility into heart health beyond clinical settings. Yet this continuous surveillance comes with an unexpected cost: the burden of false alarms.
The problem is staggering. Historically, approximately 75% of implantable loop recorder alerts are false positives, requiring significant time and effort for adjudication. Mobile cardiac telemetry devices, designed with intentionally lenient algorithms to avoid missing life-threatening events, generate a high burden of false alarms that demand manual review by technicians—increasing workload, reducing efficiency, and creating unreimbursed labor costs. For smartwatch-based detection systems, false alarms can reach patients directly, creating anxiety and eroding trust in the technology.
This is the false alarm paradox: devices designed to protect patients risk overwhelming both clinicians and wearers with noise, potentially causing them to ignore alerts entirely—a phenomenon known as “alert fatigue.” Solving this challenge is essential for the long-term viability of remote cardiac monitoring.
The Human Impact: When False Alarms Harm
The consequences of false alarms extend far beyond clinical inefficiency.
Analysis from the Pulsewatch study — examining stroke survivors using smartwatches for AF detection — revealed a troubling dose-dependent relationship: receipt of false AF alerts was associated with a decline in self-perceived physical health and levels of disease self-management confidence. The more false alarms participants received, the worse they felt about their health and their ability to manage their condition.
Key Findings from the Pulsewatch Study
- Among 85 stroke survivors randomized to receive smartwatch monitoring, 15 received AF alerts.
- 10 of those 15 participants (67%) received alerts that were false positives — no AF was detected on contemporaneous ECG patch monitoring.
- Of the 35 false alerts analyzed:
- 19 occurred during sinus rhythm with noisy PPG signals
- 11 were triggered by benign arrhythmias like premature atrial or ventricular contractions
This psychological impact matters. Patients prescribed cardiac monitors are often already anxious about their heart health. False alarms amplify that anxiety while simultaneously undermining confidence in the technology meant to protect them. When patients lose trust in their monitors, adherence suffers, and the entire purpose of remote monitoring collapses.
The Clinical Burden: Alert Fatigue and Resource Drain
For healthcare providers, false alarms create an unsustainable workload. Independent Diagnostic Testing Facilities (IDTFs) reviewing mobile cardiac telemetry data must manually evaluate each alert, with false positives consuming time that could be spent on genuine clinical concerns. The economic impact is equally severe: unreimbursed labor for reviewing false alerts strains already tight margins.
The phenomenon of alert fatigue is well-documented: when clinicians are bombarded with notifications, they begin ignoring them. A case study from Adamo Software describes a remote patient monitoring system where clinicians disabled notifications entirely because the wearable heart monitor generated numerous false alarms due to movement artifacts. This is the nightmare scenario: a system designed to protect patients becomes so noisy that clinicians tune it out entirely.
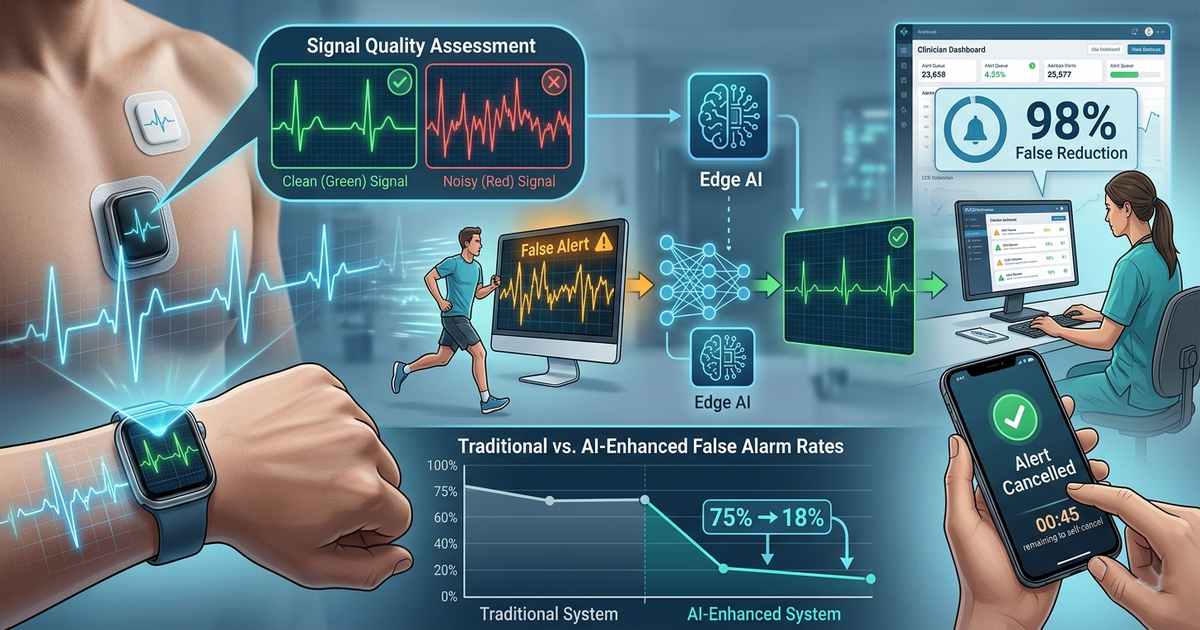
Signal Quality Assessment: The First Line of Defense
The most direct approach to reducing false alarms is preventing them at the source by ensuring only high-quality signals are analyzed. Recent research from IIT Indore presents a breakthrough: derivative ECG (dECG) signal-based lightweight convolutional neural networks (CNNs) for automatic ECG signal quality assessment.
CNN Performance Results
- 97.59% accuracy on unseen ECG databases
- 98.78% sensitivity
- 89.23% specificity
- 2,989 kB model size — suitable for resource-constrained devices
- 130.44 ± 46.24 milliseconds processing time for 5-second ECG signal on Raspberry Pi
This lightweight approach is essential. Wearable devices have limited computing capacity and onboard memory. By embedding signal quality assessment directly on the device, only clinically interpretable data triggers alerts, dramatically reducing false positives while maintaining sensitivity to genuine arrhythmias.
Advanced AI: Learning to Ignore Noise
Beyond basic signal quality assessment, sophisticated AI algorithms are learning to distinguish artifacts from genuine pathology. The Pulsewatch study developed a novel convolutional denoising autoencoder (CDA) approach that fundamentally rethinks artifact removal.
Dual-CDA Architecture Innovation
- Traditional approach: Single CDA trained on normal sinus rhythm applied to all data — suboptimal for AF episodes
- Pulsewatch innovation: Two distinct CDA models trained separately on AF and non-AF data
- Process: Deep learning classifier determines rhythm type → routes to appropriately trained CDA model → targeted artifact removal
For atrial fibrillation detection specifically, research from Chalmers University reveals a critical trade-off. Smartwatch algorithms designed to minimize false positives may compromise sensitivity for sparse or intermittent AF. Analysis showed that for individuals with low-to-moderate AF burden (0.5%–1.5%), over 20% of cases remained undetected after five years of monitoring. This finding underscores that false alarm reduction must balance specificity without sacrificing detection of genuine — but intermittent — pathology.
Hardware Innovation: Better Sensors, Fewer Artifacts
Signal processing algorithms are only as good as the data they receive. Hardware advances are reducing artifacts at the source. Nanochap’s BAF003 chip, designed for integration into smart clothing, addresses the fundamental limitations of wrist-based monitoring.
BAF003 Chip Specifications
- 120x programmable gain amplifier — captures ECG fluctuations as low as 0.5mV
- 2kHz high-speed ADC — sampling at 2000 samples per second
- 91% motion interference suppression — compared to 23% false alarm rate in traditional chips
By embedding sensors directly into fabric rather than wristbands, these systems maintain skin contact even during movement and sleep, dramatically reducing the artifact burden that triggers false alarms.
The Grace Period: Human-in-the-Loop Validation
Some false alarms can only be identified by the wearer themselves. Research from Eindhoven University of Technology explores a simple but powerful intervention for smartwatch-based out-of-hospital cardiac arrest detection: the grace period.
Grace Period Study Results
- 10-second grace period: 98.3% of (audio)tactile alarms would be canceled by users
- 20-second grace period: 99.6% of (audio)tactile alarms would be canceled by users
- (Audio)tactile alarms significantly shortened response times compared to auditory-only alarms
For life-threatening events like cardiac arrest, a 10-20 second delay before EMS notification is clinically insignificant. For false alarms, this grace period allows wearers to cancel erroneous alerts before they trigger emergency response — reducing unnecessary emergency dispatches while maintaining safety.
Remote Reprogramming: Adaptive Algorithms Over Time
Implantable loop recorders present unique challenges. These devices monitor for years, and patient physiology or arrhythmia patterns may evolve. The VA National Cardiac Device Surveillance Program studied patients with LINQII and LUX-Dx implantable loop recorders — devices featuring dual-stage AI algorithms and remote reprogramming capability.
Remote Reprogramming Results
- LINQII devices: Reprogrammed 22 times — alerts per day reduced from 0.13 to 0.03
- LUX-Dx devices: Reprogrammed 52 times — alerts per day reduced from 0.15 to 0.01
- Clinical outcome: No adverse events that could have been identified by superior or earlier arrhythmia detection
Remote reprogramming enables clinicians to titrate alert sensitivity based on observed false positive patterns, patient tolerance, and changing clinical needs — all without device replacement or office visits.
Edge AI: Processing at the Source
The most comprehensive approach to false alarm management shifts intelligence to the device itself. Adamo Software’s work with an Australian remote patient monitoring partner demonstrates the edge-first philosophy.
Edge AI Pipeline
- Filter: Remove non-clinical artifacts like arm movement
- Analyze: Detect trends deviating from patient-specific baseline
- Transmit: Send only meaningful events to the cloud
Results
- 60% reduction in data storage costs (from ~4 terabytes per device)
- Restored clinician trust: “when the dashboard alerts, doctors now pay attention”
This approach recognizes a fundamental truth: medical value lies not in data volume but in clinical insight. The best monitoring system isn’t the one capturing the most data — it’s the one generating the most meaningful alerts.
The Path Forward: Multi-Layered False Alarm Management
No single intervention solves the false alarm problem. The future lies in multi-layered strategies combining multiple approaches:
- At the sensor layer: Advanced hardware like the BAF003 chip suppresses motion artifacts at the source, reducing false alarm triggers before they occur.
- At the signal processing layer: Lightweight CNNs assess signal quality in real-time, discarding noisy segments before analysis.
- At the AI layer: Specialized models — like dual-CDA architectures — distinguish artifacts from pathology with increasing sophistication.
- At the human interface layer: Grace periods empower wearers to cancel false alarms before they reach emergency services.
- At the system layer: Remote reprogramming enables adaptive algorithms that evolve with patient needs.
- At the cloud edge: Edge AI filtering ensures only clinically meaningful events reach clinicians.
Conclusion
Remote cardiac monitoring wearables represent one of medicine’s most transformative technologies. Yet their long-term viability depends on solving the false alarm problem — not just for clinical efficiency, but for patient trust and well-being.
The evidence is clear: false alarms harm patients, overwhelm clinicians, and undermine the very purpose of monitoring. But equally clear is the path forward. Through signal quality assessment, sophisticated AI, hardware innovation, grace periods, remote reprogramming, and edge processing, we can build systems that alert only when necessary — and are trusted when they do.
The goal isn’t eliminating all false alarms — that’s impossible while maintaining sensitivity to genuine events. The goal is reducing them to a level where clinicians and patients can confidently respond when it matters most.